What is water-based polyurethane resin?
The first polyurethane was discovered by Autobayer in Germany in 1937, and after that, these materials made great progress in all kinds of industries in the world due to their special properties.
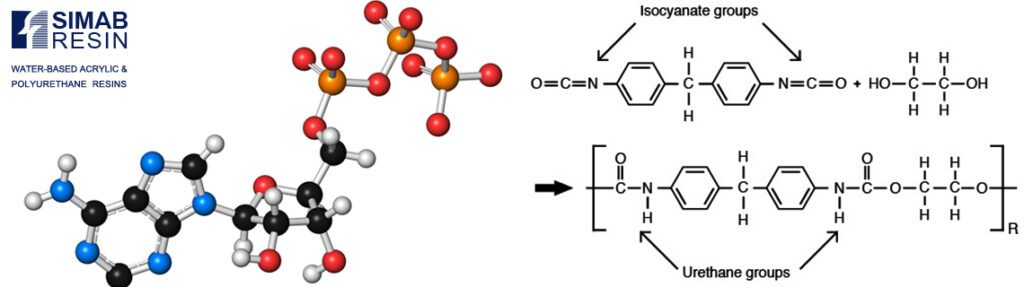
Polyurethane (PU) is the general name of resins that have urethane bonds. The urethane bond is formed through an addition reaction between an isocyanate group and a compound with an active hydrogen such as a hydroxyl group. Isocyanate groups are highly reactive, and for this reason, the progress of their reaction does not require an increase in temperature. (The reaction takes place at room temperature).
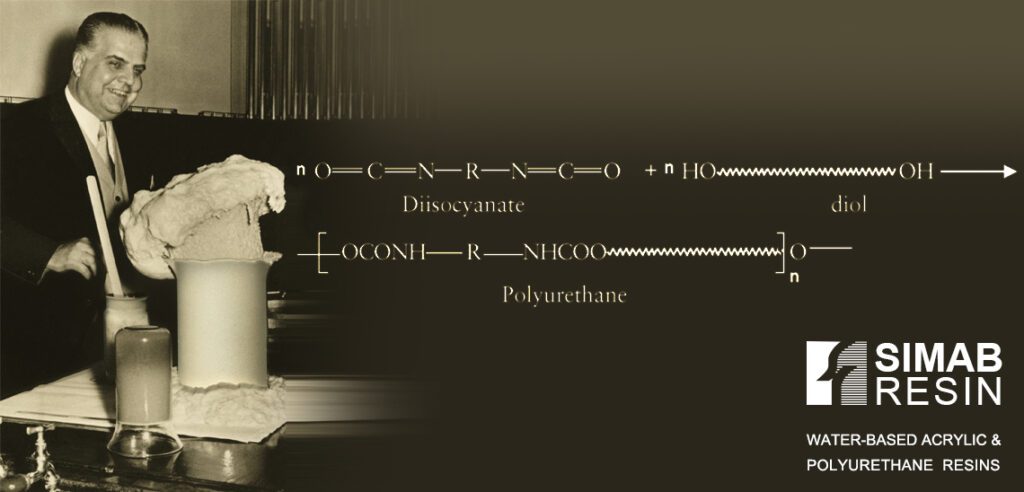
What are the different forms of water-based polyurethane resin?
Polyurethane can be produced in different forms such as: foam, film, elastomers, powders, liquids and emulsions. Compounds that have an isocyanate group are:
2.4 or 2.6 toluene diisocyanate
4.4 or 2.4 diphenylmethane diisocyanate
1.6 hexamethylene diisocyanate

What is polyol?
In addition to the ones mentioned, there are other isocyanate compounds.
Compounds that have more than two hydroxyl groups (OH) are called polyols, and the following species are usually used in polyurethane synthesis:
Polyether polyol
Polyester polyal
Polycarbonate Polyol
Polycaprolactone Polyl
What compounds with isocyanates can be used?
In addition, instead of hydroxyl groups, compounds such as carboxylic acids and amines, which have active hydrogen, can also be used in combination with isocyanates. If, instead of di-alcohol, a diamine reacts with isocyanate, the resulting resin is placed in the polyurea family. Spandex is a composite with a complex structure that includes both urea and urethane functional groups. Therefore, the variety of reagents that are used against isocyanates leads to the production of polyurethanes in various types with different characteristics. For this reason, when it comes to polyurethanes, it can be said that there are countless types of them.
Introduction of water-based polyurethane dispersions (PUD)
Waterborne Polyurethane Dispersions (PUD) is a type of waterborne polyurethane that was first made in the 1960s by dispersing polyurethane particles in water. This coating can be a suitable alternative to solvent-based coatings due to its greater compatibility with the environment and lower amount of volatile organic compounds (VOC). PUDs are used in a variety of applications including wood coatings, automotive coatings, and textile finishes. They have excellent durability, chemical resistance and good UV stability. In addition, they are easy to use and dry quickly.
Primary water-based polyurethane dispersions were made by dispersing polyurethane prepolymers in water using surfactants and stabilizers. These early PUDs were initially used as adhesives and coatings.
In the 1970s, advances in polymer chemistry led to the development of more complex water-based polyurethane dispersions with improved properties such as increased durability, flexibility, and chemical and abrasion resistance. These new water-based polyurethanes are used in a wide range of industries including automotive, textile, leather and wood coatings.
In the 1980s, environmental concerns led to increased interest in water-based coatings and adhesives. PUDs have quickly become popular due to offering many of the same benefits as solvent-based coatings without the environmental disadvantages.
Recent advances in waterborne polyurethane dispersion technology have focused on improving their performance characteristics such as scratch resistance, UV stability, and water resistance. In addition, efforts are underway to develop more sustainable PUD formulations that use renewable raw materials and have lower environmental impacts.

What are the applications of water-based polyurethane dispersions (PUD)?
In general, water-based polyurethane dispersions (PUD) is a widely used material that is used in various industries due to its excellent properties such as adhesion, durability, flexibility, water resistance, chemical resistance, UV resistance, etc. Some common uses of PUD include:
Coatings: PUDs are used as coatings for various substrates such as wood, metal, plastic, and textiles. They have excellent adhesion, durability and resistance to abrasion, chemicals and UV rays.
Adhesives: PUDs are used as adhesives for gluing different materials such as wood, paper, leather and textiles. The strength and flexibility of these materials is high.
Textile finishing: PUDs are used in textile finishing to provide properties such as water repellency, stain resistance, and wrinkle resistance.
Leather finishing: PUDs are used in leather finishing to improve the appearance of leather by providing a glossy or matte finish.
Inkjet printing: PUDs are used in inkjet printing to produce high quality prints on various substrates such as paper, plastic films and textiles.
Automotive Coatings: PUDs are used in automotive coatings to provide adequate scratch resistance and durability.
Construction: PUDs are used in construction applications such as sealants and waterproofing membranes due to their excellent adhesion properties and water resistance.
Packaging: PUDs are used in packaging applications such as laminated films for food packaging due to their excellent barrier properties against oxygen and moisture.
What are the advantages of water-based polyurethane?
Water-based polyurethane dispersions (PUD) are increasingly popular due to their environmental compatibility and low volatile emissions. Here are some of the advantages of water-based polyurethane dispersions over solvent-based polyurethane:
Environmental friendliness: PUD is water-based, meaning it has less environmental impact than solvent-based coatings. It also has lower VOC emissions, making it safer for workers and the environment.
Better adhesion: Water-based polyurethane has better adhesion to all types of substrates than solvent-based coatings. This means it can be used on a wider range of surfaces, including plastic and metal.
Faster drying time: Water-based polyurethane dries faster than solvent-based coatings, making it faster and more efficient to apply.
Improved Durability: PUD is more resistant to chemicals than solvent-based coatings and is suitable for use in high-traffic areas or environments exposed to chemicals.
A disadvantage of waterborne polyurethane dispersions (PUD) is that they can be sensitive to moisture and may degrade over time when exposed to excessive moisture or water. In addition, it may have less wear resistance than other coatings.
